A group of researchers from the Okinawa Institute of Science and Technology created perovskite solar panels that maintained high efficiencies for over 1000 hours. The dimensions of the modules were 5×5 sq. cm. and 10×10 sq.cm., sizes which are much larger than the ones traditionally made in laboratories (1.5×1.5 sq. cm.). Although these solar panels are smaller than the commercially available ones, the scientists from OIST have claimed that they have improved the module’s stability and efficiency by mixing precursor materials with ammonium chloride during fabrication.
OIST Perovskite Solar Module Research Team
Their study and findings were published in Advanced Energy Materials on the 25th of January this year of 2021. It has the title: “Scalable Fabrication of >90 cm2 Perovskite Solar Modules with >1000 h Operational Stability Based on the Intermediate Phase Strategy”
- Guoqing Tong
- Dae‐Yong Son
- Luis K. Ono
- Yuqiang Liu
- Yanqiang Hu
- Hui Zhang
- Afshan Jamshaid
- Longbin Qiu
- Zonghao Liu
- Yabing Qi
Materials
With regards to their experiments, they have used the following reagents.
- Methylammonium iodide (MAI), methylammonium bromide (MABr)
- methylammonium chloride (MACl) and formamidinium iodide (FAI)
- Lead iodide (PbI2, 99.99%)
- Cesium iodide (CsI, 99.999%)
- ammonium chloride (NH4Cl, 99.5%)
- 2,2′,7,7′-tetrakis (N,N-di-p-methoxyphenylamine)-9,9′-spirobifluorene (spiro-OMeTAD)
- N,N-dimethylformamide (DMF),
- isopropanol (IPA) chlorobenzene
- 4-tert-butylpyridine (99.9%)
- acetonitrile (99.9%)
- Commercial tin (IV) oxide solution (SnO2, 15% in H2O colloidal dispersion)
Roadblocks to Perovskite Solar Modules Development
Within almost a decade, perovskites prove to be the most promising and game-changing materials for the future of solar energy technology. Perovskite’s efficiency has greatly soared from 4% to a record-breaking 29.15% over the past few years. Although it is a remarkable feat that perovskite solar cells can be produced at a cheaper price, they still encounter 2 main roadblocks to their mass production. One is their questionable stability for a long-term period, and the other thing is the huge challenge with upscaling. The perovskite cell’s promising potential to be increasingly versatile is one of the reasons the researchers strive to overcome these difficulties.
According to Dr. Guoqing Tong, one of the OIST researchers, “Perovskite material is fragile and prone to decomposition, which means the solar cells struggle to maintain high efficiency over a long time. And although small-sized perovskite solar cells have a high efficiency and perform almost as well as their silicon counterparts, once scaled up to larger solar modules, the efficiency drops.”
Perovskite Layers
How does a perovskite layer look like? It can be found within the central part of a solar device. Two other layer types are sandwiching the perovskite material, namely the electrode and the charge carrier layers. As soon as sunlight beams into the solar device, the perovskite layer absorbs it and produces charge carriers. This process generates direct current when charge carriers start to flow to the electrodes through the transport layers.
However, some factors including pinholes and material defects found between perovskite grains disrupt the flow of current within perovskite solar devices. These constraints eventually reduce the efficiency and lifespan of the perovskite layer, thus, degrading the solar device as a whole.
Perovskite Researchers Aim to Bring Solutions
Dr. Tong explained that there is a huge challenge in scaling up perovskite solar modules, “…as the modules increase in size, it’s harder to produce a uniform layer of perovskite, and these defects become more pronounced,” he added. The research team aims to find a solution to make the mass-production of perovskite solar panels feasible.
The researchers have found out that more defects and pinholes are often developed when manufacturing larger solar panels with perovskite layer. It was noted that most solar cell manufacturers today use a perovskite layer with only 500 nanometers in thickness. This is why Dr. Tong and his teammates opted to produce solar modules having perovskite films with 1,000 nanometers in thickness.

They have added ammonium chloride to solve the issues on the solubility of lead iodine which also allows the material to be uniformly dissolved. This resulted in the production of a perovskite film with fewer defects and bigger grains. They later removed the ammonia from the perovskite solution to lower the level of impurities within the film.
Results
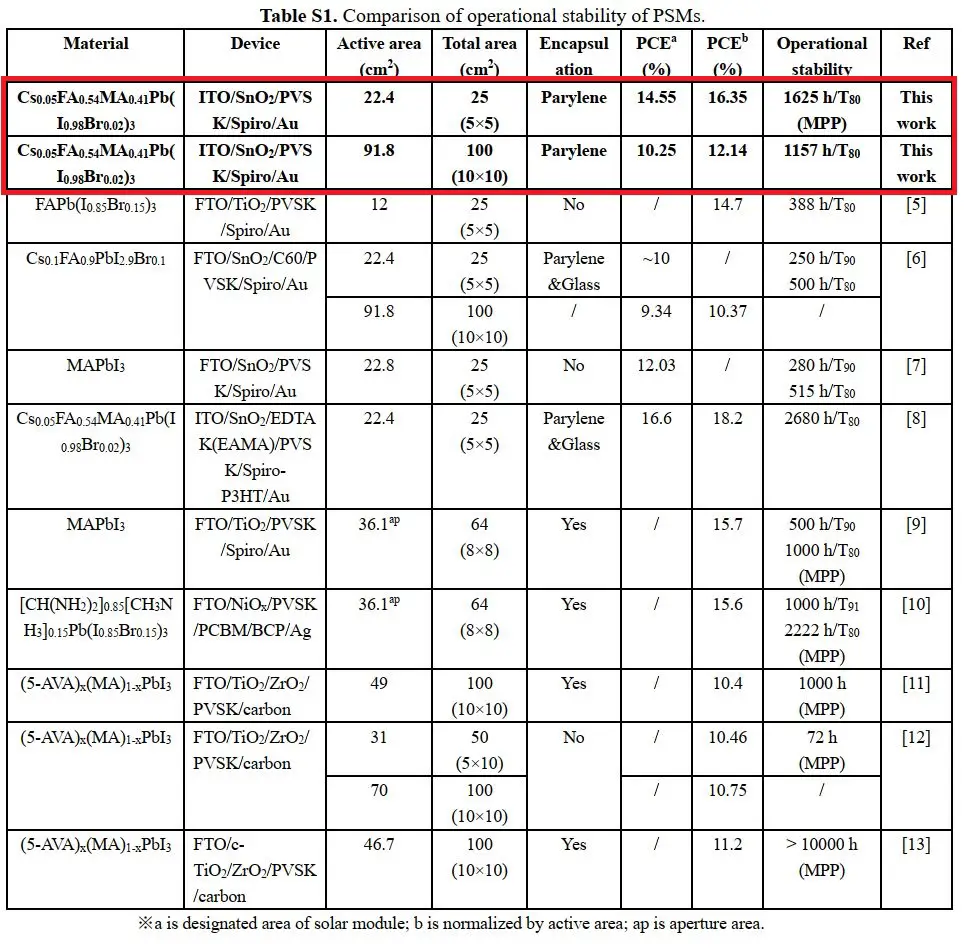
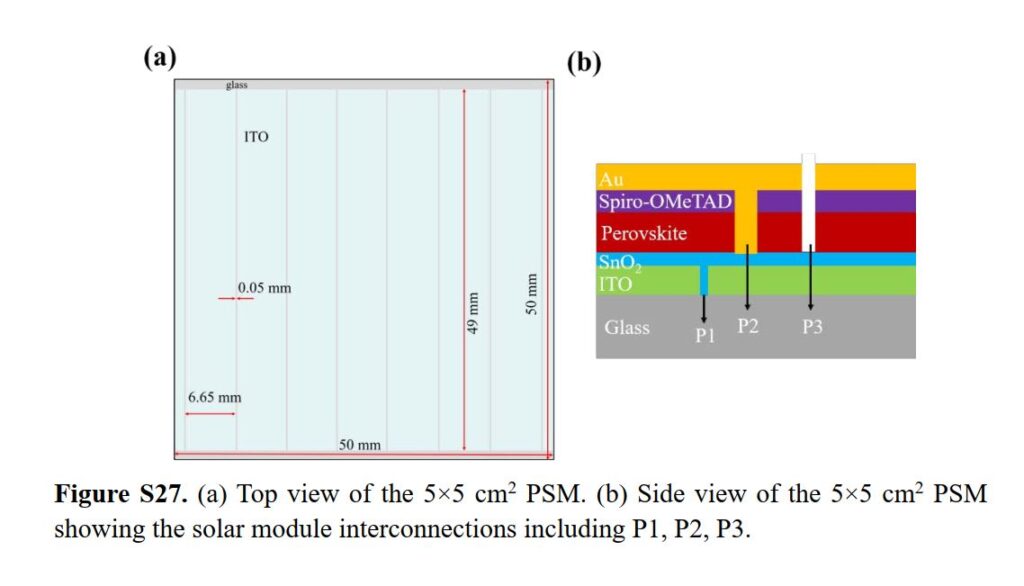
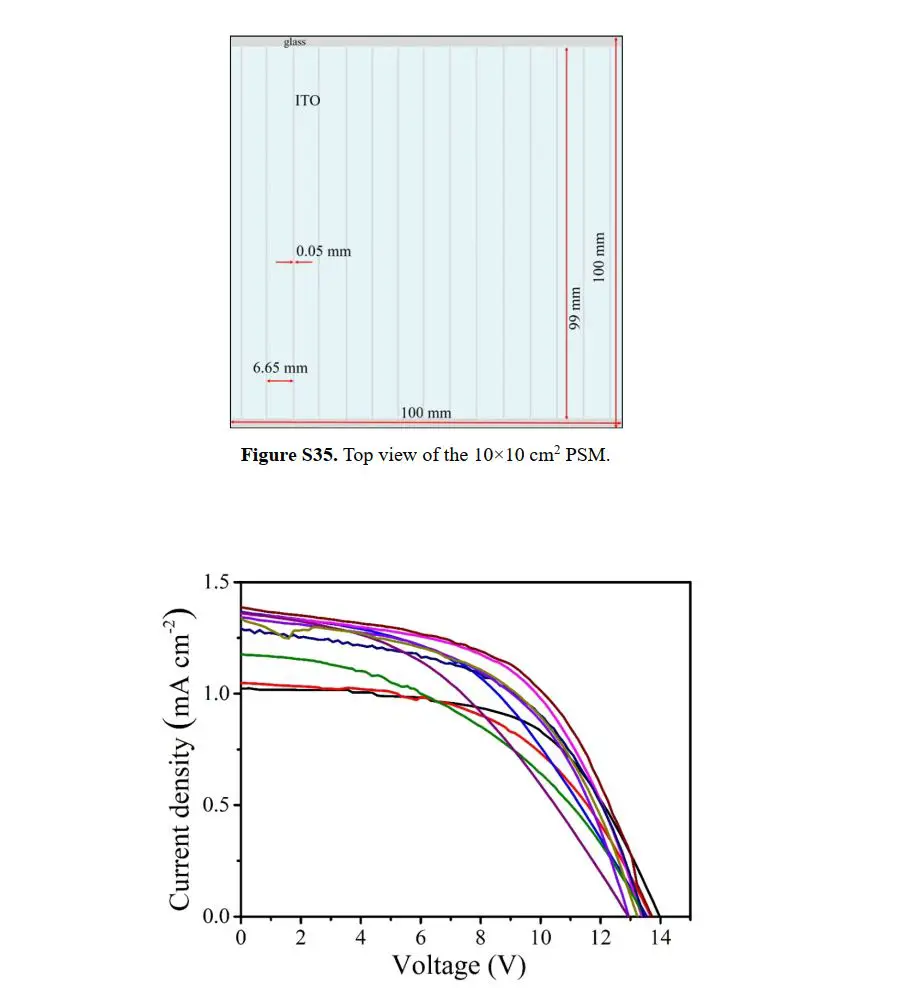
As a result, the team has recorded the data as represented by the table above wherein there is a noticeable increase in terms of stability of their perovskite solar films compared to previously studied materials. This research was published by OIST with the title, “Scalable Fabrication of >90 cm2 Perovskite Solar Modules with >1000 h Operational Stability Based on the Intermediate Phase Strategy”. The said research was supported by the OIST Technology Development and Innovation Center’s Proof-of-Concept Program.
The experiment is yet another great breakthrough towards the development of perovskite solar modules. As Dr. Tong has mentioned, “Going from lab-sized solar cells to 5 x 5 cm2 solar modules was hard. Jumping up to solar modules that were 10 x 10 cm2 was even harder. And going to 15 x 15 cm2 solar modules will be harder still”. Thankfully, the team of researchers from OIST is looking forward to the challenge.
References
- “Scalable Fabrication of >90 cm2 Perovskite Solar Modules with >1000 h Operational Stability Based on the Intermediate Phase Strategy” DOI: 10.1002/aenm.202003712
- [1] Z. Liu, L. Qiu, E. J. Juarez-Perez, Z. Hawash, T. Kim, Y. Jiang, Z. Wu, S. R. Raga, L. K. Ono, S. F. Liu, Y. B. Qi, Nat. Commun. 2018, 9, 3880.
- [2] G. Tong, H. Li, G. Li, T. Zhang, C. Li, L. Yu, J. Xu, Y. Jiang, Y. Shi, K. Chen, Nano Energy 2018, 48, 536.
- [3] E. J. Juarez-Perez, Z. Hawash, S. R. Raga, L. K. Ono, Y. B. Qi, Energy Environ. Sci. 2016, 9, 3406.
- [4] E. J. Juarez-Perez, L. K. Ono, M. Maeda, Y. Jiang, Z. Hawash, Y. B. Qi, J. Mater. Chem. A 2018, 6, 9604.
- [5] Y. Jiang, M. Remeika, Z. Hu, E. J. Juarez‐Perez, L. Qiu, Z. Liu, T. Kim, L. K. Ono, D. Y. Son, Z. Hawash, M. R. Leyden, Z. Wu, L. Meng, J. Hu, Y. B. Qi, Adv. Energy Mater. 2019, 9, 1803047.
- [6] L. Qiu, S. He, Y. Jiang, D.-Y. Son, L. K. Ono, Z. Liu, T. Kim, T. Bouloumis, S. Kazaoui, Y. B. Qi, J. Mater. Chem. A 2019, 7, 6920.
- [7] L. Qiu, Z. Liu, L. K. Ono, Y. Jiang, D. Y. Son, Z. Hawash, S. He, Y. B. Qi, Adv. Funct. Mater. 2018, 29, 1806779.
- [8] Z. Liu, L. Qiu, L. K. Ono, S. He, Z. Hu, M. Jiang, G. Tong, Z. Wu, Y. Jiang, D.- Y. Son, Y. Dang, S. Kazaoui, Y. B. Qi, Nat. Energy 2020, 5, 596.
- [9] H. Chen, F. Ye, W. Tang, J. He, M. Yin, Y. Wang, F. Xie, E. Bi, X. Yang, M. Gratzel, L. Han, Nature 2017, 550, 92.
- [10] E. Bi, W. Tang, H. Chen, Y. Wang, J. Barbaud, T. Wu, W. Kong, P. Tu, H. Zhu, X. Zeng, J. He, S.-i. Kan, X. Yang, M. Grätzel, L. Han, Joule 2019, 3, 2748.
- [11] Y. Hu, S. Si, A. Mei, Y. Rong, H. Liu, X. Li, H. Han, Sol. RRL 2017, 1, 1600019.
- [12] A. Priyadarshi, L. J. Haur, P. Murray, D. Fu, S. Kulkarni, G. Xing, T. C. Sum, N. Mathews, S. G. Mhaisalkar, Energy Environ. Sci. 2016, 9, 3687.


















